
Contact
Email: rbrinson@umd.edu
Call: (240) 314-6336
Robert Brinson
Research Chemist
Brinson Group
Contact
Email: rbrinson@umd.edu
Call: (240) 314-6336
Education
- Postdoctoral Fellow, National Cancer Institute, 2008
- Postdoctoral Fellow, Joint National Institute of Standards and Technology and National Institutes of Health National Research Council, 2006-2007
- Ph.D., Chemistry, Wake Forest University, 2005
- B.A., Chemistry and Music, Houghton College, 2001
Profile
Dr. Robert Brinson uses biophysical measurements to define the structure and dynamics of nucleic acids, proteins, and glycoproteins, with a specific focus on biotherapeutics. In particular, he seeks to harmonize and to translate nuclear magnetic resonance (NMR) methods for biopharmaceutical applications and to push the limits of NMR methodologies to define the critical quality attributes of higher order structure for protein drug products.
CURRENT RESEARCH
2D-NMR Method Development for Biopharmaceutical Structural Assessment
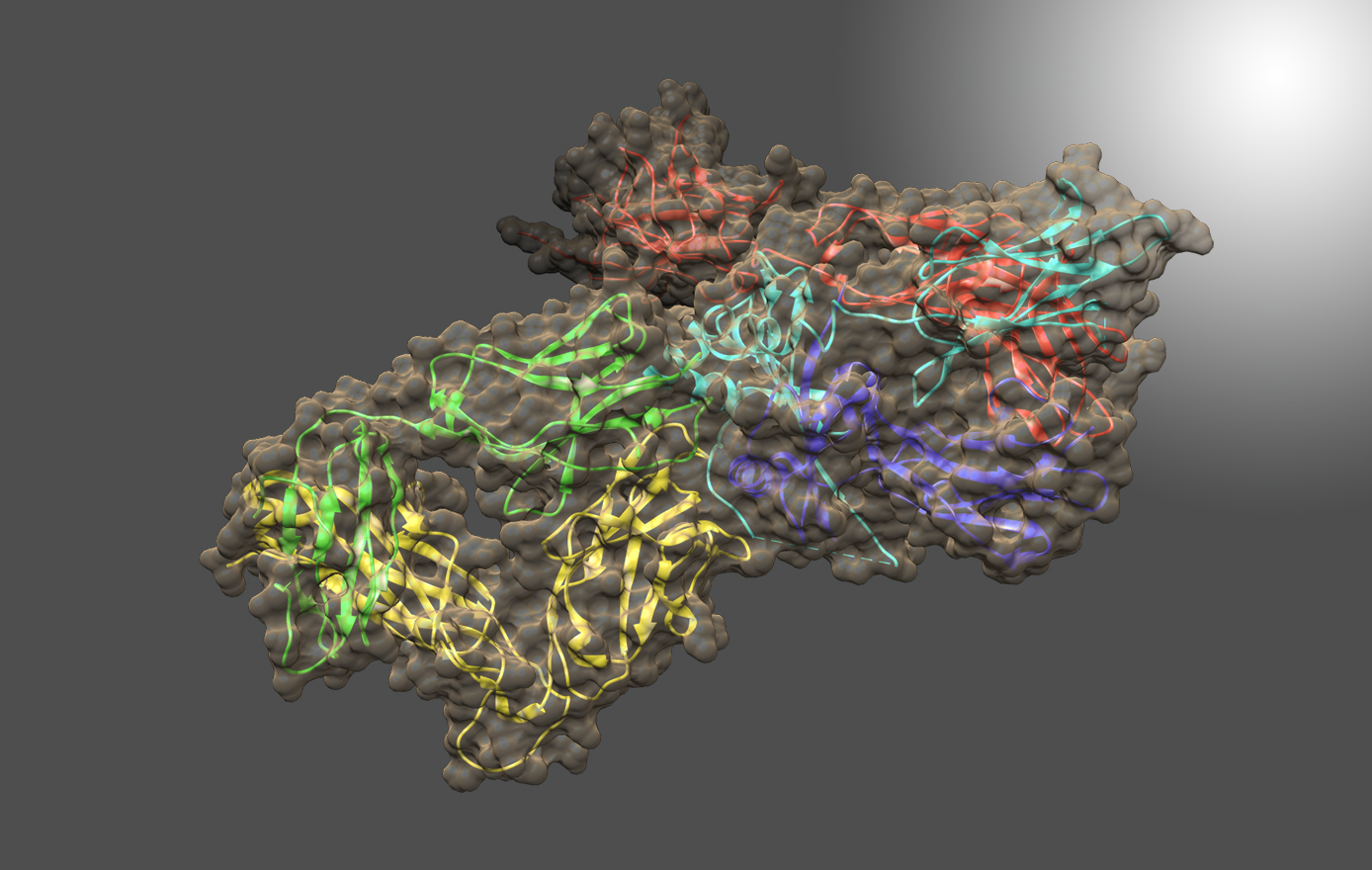
Techniques for the facile characterization of higher order structure of biotherapeutics are in great demand by the pharmaceutical industry. NMR spectroscopy provides structural information about proteins at atomic resolution as individual peak positions in ‘fingerprint’ amide or methyl NMR spectra are sensitive to structure. These peaks can serve as spectral indicators for establishing consistency in drug manufacturing, assessing the stability of drug formulations, detecting process-related drug product variations, and determining biosimilarity to innovator reference products. However, limits on sensitivity, molecular size, sample volumes, and the perceived need for stable-isotope enrichment have restrained widespread use of NMR in the biopharmaceutical industry. Recent advances in NMR hardware, acquisition methods, and new approaches for data analyses have greatly ameliorated these limitations. Two-dimensional (2D) 1H-13C and 1H-15N correlated spectra can now be collected on biologics, including monoclonal antibodies (mAbs), at natural isotopic abundance. Furthermore, in collaboration with the Mathematical Analysis and Modeling Group at NIST, Dr. Brinson develops chemometric analyses for the extraction of meaningful information from many 2D spectra. Together, the researchers aim to develop best practices based on the type of analyses needed for a given protein therapeutic sample set.
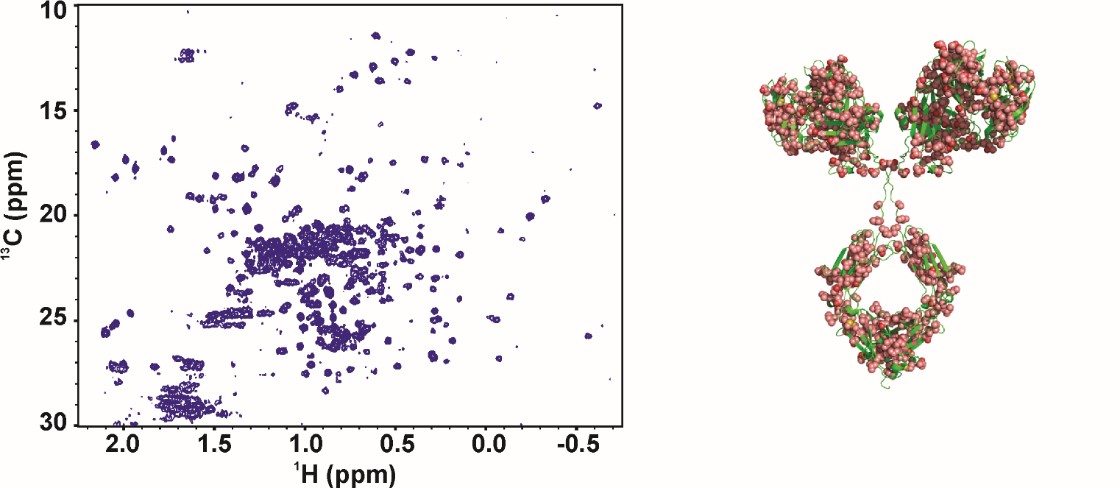
Harmonization of the 2D-NMR method through an International Interlaboratory Study
The NISTmAb Interlaboratory NMR Study is a joint effort among 25 organizations to evaluate the performance of 2D heteronuclear NMR for use in characterizing the high order structure of proteins, with the aim of supporting the use of NMR to characterize biologic therapeutics. Researchers in nine countries, with equal representation from industrial, governmental, and academic laboratories, acquired 451 1H-13C-methyl and 1H-15N-amide 2D NMR correlation spectra using different acquisition methods and instruments. Spectra were collected on both the Fab antibody domain derived from the IgG1κ NIST monoclonal antibody (NISTmAb) and a U-15N, 20%-13C labeled NIST-Fab system suitability sample. Chemometric analyses, using multivariate statistical methods, allowed benchmarking of the precision and robustness of the NMR measurements. The complete data package is available at www.ibbr.umd.edu/groups/nistmab-nmr.