Molecular machines
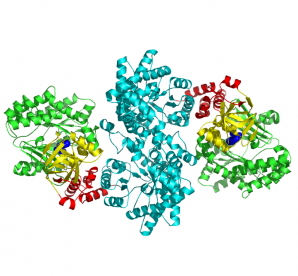
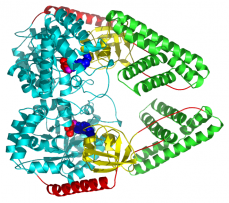
Two PEP-utilizing enzymes function in vastly different biological contexts, yet both catalyze phosphoryl group transfer by shuttling a phosphohistidine residue between remote active centers. These enzymes utilize a swivel domain mechanism to deliver the phosphoryl group to the appropriate substrate.
Pyruvate phosphate dikinase (PPDK), catalyzes the interconversion of PEP, AMP and PPi to pyruvate, ATP and Pi. The PEP/pyruvate bind in a site remotely located from the nucleotide and phosphate binding site. Enzyme I of the bacterial PEP:sugar phosphotransferase system (PTS), transfers the phosphoryl group of PEP to the next PTS protein, HPr. HPr~P then proceeds to phosphorylate a sugar specific permease responsible for the uptake of the incoming sugar. Again, PEP and HPR bind remotely from one another.
To view and download videos that depict the swiveling motions of PPDK and Enzyme I return to my home page.