Round Robin Study Supports Adoption of Targeted Attribute Analytical Method by Biopharma
Mon, Oct 10, 2022
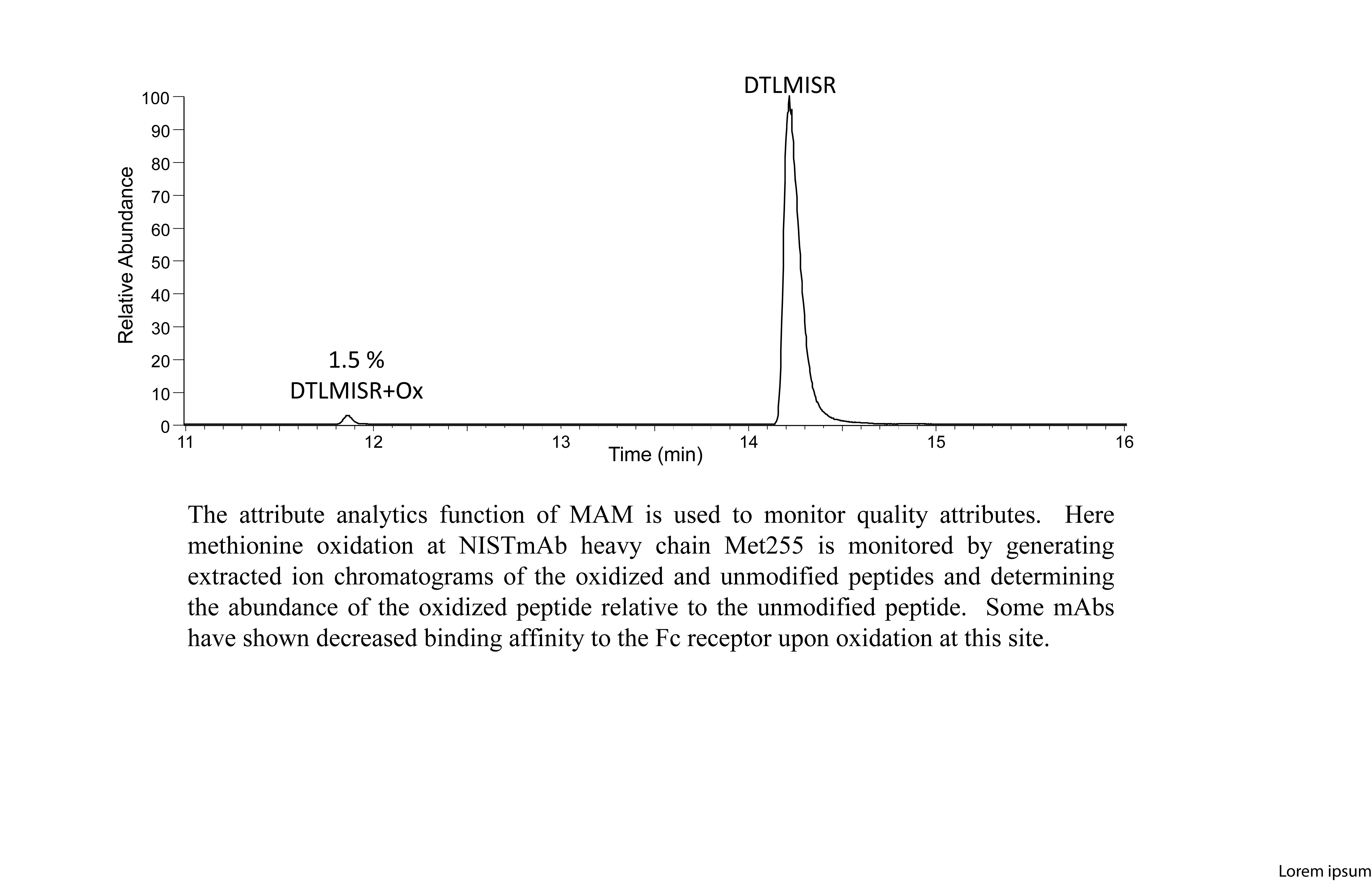
Drs. Trina Mouchahoir (NIST/IBBR) and John Schiel (NIST/IBBR Fellow) recently reported on results from a multi-attribute method (MAM) round robin study they lead together with Rich Rogers (Just - Evotec Biologics). MAM is a mass spectrometry-based measurement platform developed with an eye toward improving quality control (QC) analytics for therapeutic proteins. The study relied upon members of the MAM Consortium for participation and represented the efforts of 28 laboratories from the biopharmaceutical industry, instrument and software vendors and federal regulators.
The published article*, which follows an earlier report†, describes current industry-wide performance of targeted attribute analytics. Targeted attribute analytics has the potential to replace multiple analytical platforms used for monitoring product quality with a single assay but has yet to achieve widespread use in the QC environment. The goal of the study was to evaluate the reproducibility of attribute analytics measurements as a way of surveying the status of method performance across the industry. The NISTmAb monoclonal antibody reference material was the model therapeutic protein analyzed using the MAM platform by participants. The results demonstrated the reproducibility of MAM and orthogonality to current methods. In addition, the study brought to light certain aspects of the method that require some fine-tuning and provided the authors the opportunity to suggest best practices for developing reproducible MAM workflows. It is anticipated that the results of this study will aid improvements to the method and further facilitate the adoption of MAM across the industry.
Along with the publications, the raw data generated by the study participants has been made available using NIST’s public data repository (https://doi.org/10.18434/mds2-2497). The availability of these data sets is projected to be a valuable tool for the development of software platforms designed for MAM data analysis.
*Mouchahoir T, Schiel JE, Rogers R, et al. Attribute Analytics Performance Metrics from the MAM Consortium Interlaboratory Study. Journal of the American Society for Mass Spectrometry. 2022, 33(9): 1659-1677. (http://pubs.acs.org/doi/abs/10.1021/jasms.2c00129).
†Mouchahoir T, Schiel JE, Rogers R, et al. New Peak Detection Performance Metrics from the MAM Consortium Interlaboratory Study. Journal of the American Society for Mass Spectrometry. 2021, 32(4): 913-928. (http://pubs.acs.org/doi/abs/10.1021/jasms.0c00415).
