Contact
Email: amackerell@rx.umaryland.edu
Call:
Alexander MacKerell
Weber Group
Contact
Email: amackerell@rx.umaryland.edu
Call:
Education
- Ph.D., Biochemistry, Rutgers University, 1985
- B.S., Chemistry, University of Hawaii, 1981
- A.S., Biology, Gloucester County College, 1979
Subsequent training involved postdoctoral fellowships in the Department of Medical Biophysics, Karolinska Intitutet, Stockholm, Sweden in experimental and theoretical biophysics and in the Department of Chemistry, Harvard University in theoretical chemistry.
Profile
Dr. Alexander MacKerell’s research involves the development and application of computational methods to investigate the relationships of structure and dynamics to function in a range of biological and chemical systems. These efforts include empirical force field development, implementation of novel sampling methodologies, understanding the physical forces driving the structure and dynamics of proteins, nucleic acids, and carbohydrates, and computer-aided drug design (CADD) studies and methodology development. The MacKerell lab works closely with experimentalists in the area of drug development to provide detailed interpretation of experimental data while simultaneously refining and developing novel theoretical approaches.
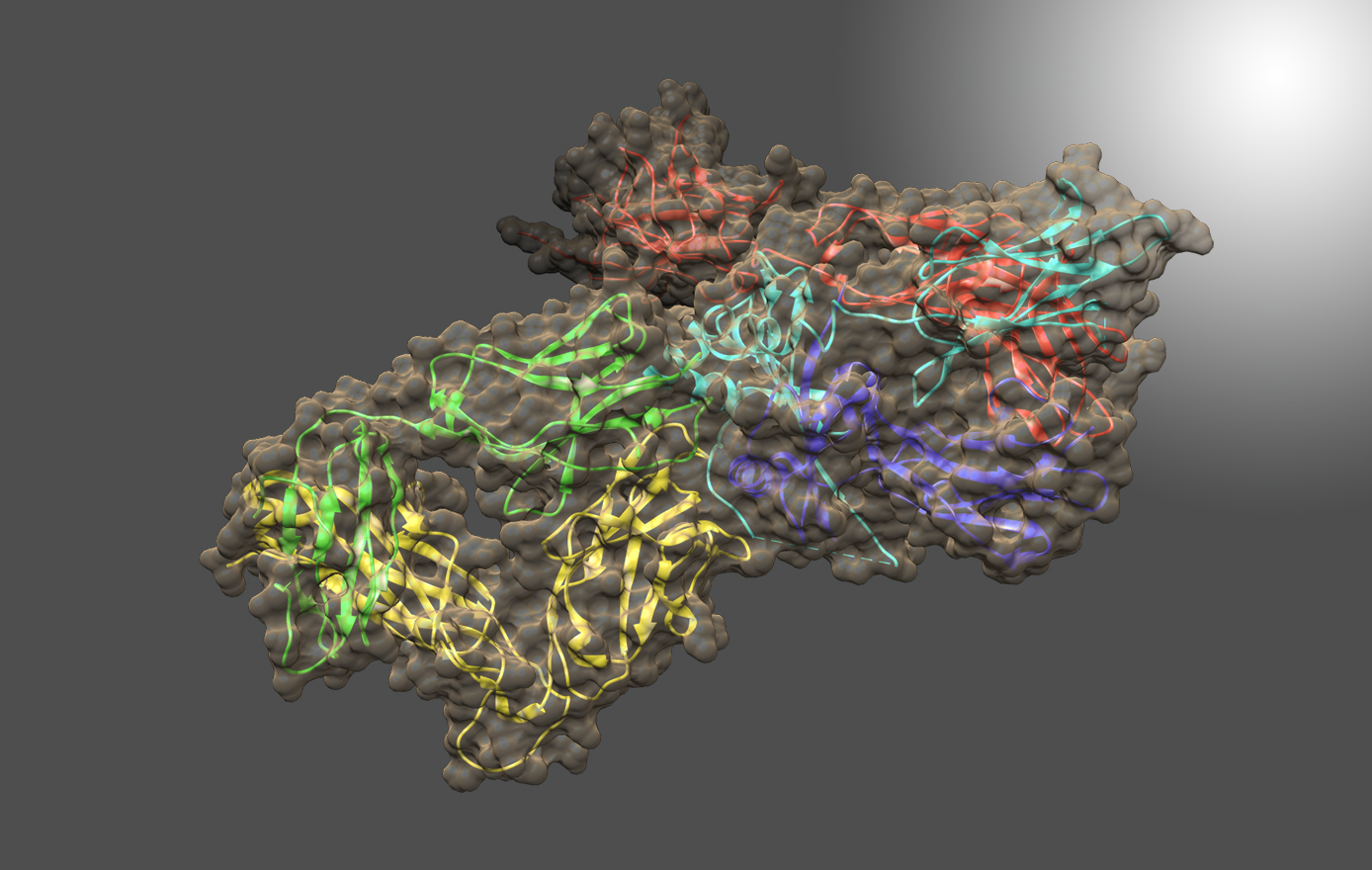
The MacKerell lab is responsible for developing and maintaining the empirical force fields used in a number of simulation packages including the program CHARMM (Chemistry at HARvard Macromolecular Mechanics). CADD methodologies developed in the MacKerell lab include SILCS (Site-Identification by Ligand Competitive Saturation) and CSP (Conformationally Sampled Pharmacophore).
CURRENT RESEARCH
A central focus of Dr. MacKerell’s group is the continued development and extension of empirical force fields and enhanced sampling methods for use in the simulation of biological macromolecules. Such simulations are important in advancing the understanding of how intrinsic characteristics and environmental conditions contribute to the conformational properties of proteins, nucleic acids, lipids, carbohydrates, and small molecules.
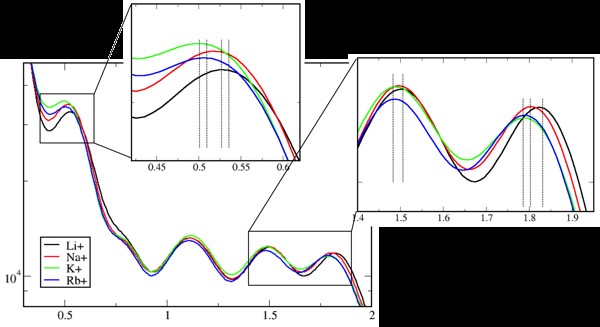
In addition to widely used additive force fields, the MacKerell lab developed a polarizable force field for biomolecules, based on a classical Drude oscillator model. Drude-2013 extends the capabilities of molecular dynamics simulations, as in the example shown in the graph to the right.
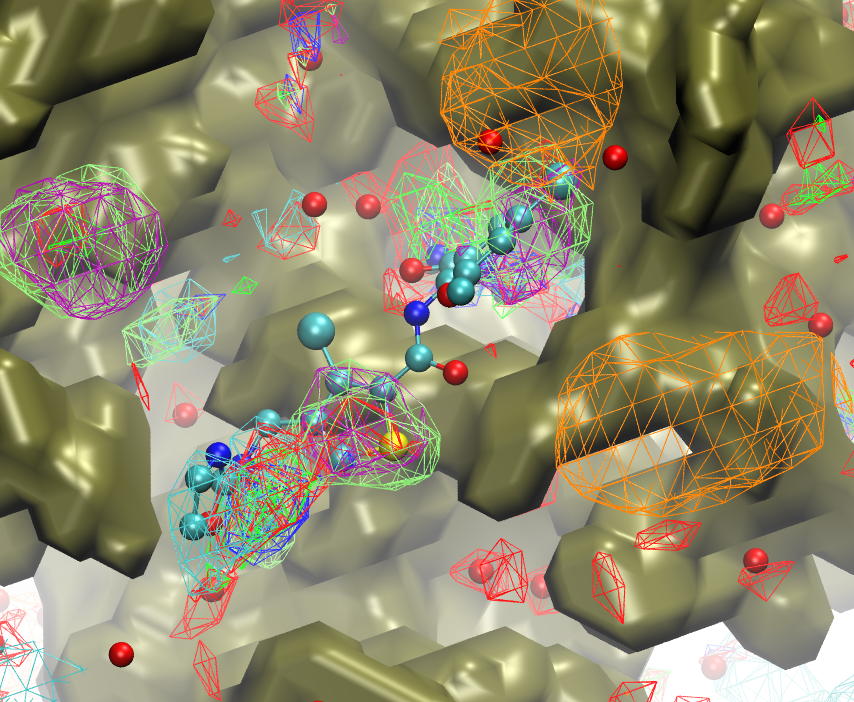
CADD is a powerful tool that must contend with both the large conformational space of flexible macromolecular drug targets like proteins and the large chemical space of potential drug-like molecules to be screened against the target. Work in the MacKerell lab seeks to address these challenges with the development of new methodologies. For example, the SILCS (Site Identification by Ligand Competitive Saturation) methodology may be used for lead compound identification and optimization, evaluation of protein-protein interactions, and optimization of formulation via rational selection of excipients and buffer.
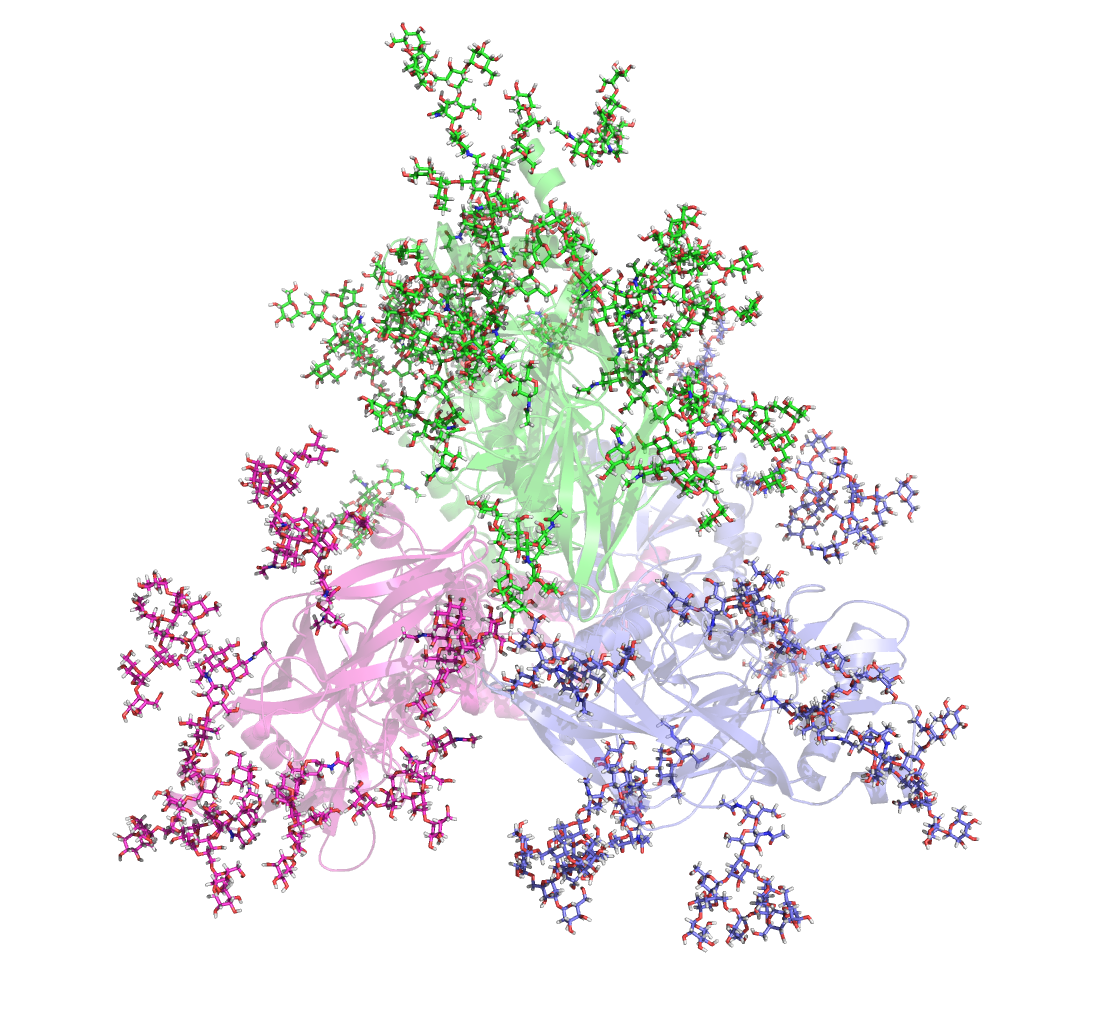
Additional activities in the MacKerell lab include structure-function studies of carbohydrates, proteins, and nucleic acids. Recently, the lab used enhanced sampling methods to explore the conformational heterogeneity HIV envelope protein glycans (Yang et al. 2017, Scientific Reports), resulting in the identification of novel glycan-antibody interactions that may play a role in neutralizing the virus. See https://mackerell.umaryland.edu/ for additional information on the MacKerell lab.