Profile

Gregory Payne
Research Professor
Payne Group (301) 405-8389 gpayne@umd.eduThe last century witnessed spectacular advances in both microelectronics and biotechnology yet there remains considerable opportunity to create synergies between the two. The Payne laboratory aims to fabricate high-performance material systems to span the capabilities of biology and information technology. Through an extensive network of international collaborations their group focuses on two primary areas of research:1) biofabrication of the bio-device interface and 2) redox-based molecular communication for bio-device "connectivity".
CURRENT RESEARCH
Biofabrication of the Bio-device Interface
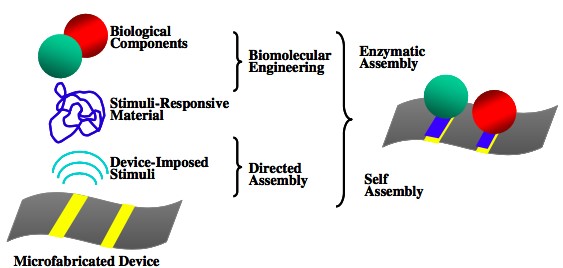
Biology is expert at creating functional nanoscale components (e.g., proteins) and assembling them over a hierarchy of length scales. However, biology’s use of labile components and bottom-up assembly differs markedly from the “bio-incompatible” top-down methods used to fabricate microelectronic devices. Biofabrication – the use of biological materials and mechanisms for construction – offers the opportunity to span these divergent fabrication paradigms by providing convergent approaches for building the bio-device interface. Figure 1 illustrates the lab’s vision for biofabricating the bio-device interface using device-imposed electrical stimuli to provide the cues to trigger biopolymers (e.g., the polysaccharide chitosan) to locally self-assemble into a hydrogel matrix at an electrode address. Importantly, the electrodeposited biopolymer hydrogels provide the water-rich microenvironment that is compatible with biology. Further, these biopolymers can be bio-functionalized through biological mechanisms (e.g., enzymes can be used to graft functional components onto these biopolymer hydrogels).
Redox-based Molecular Communication for Bio-device "Connectivity"
Biology and electronics are each expert at processing information yet they use entirely different signaling modalities. Biology and electronics are each expert at processing information yet they use entirely different signaling mechanisms. Biology signals using ions and molecules, while modern devices use electrons to process information. Oxidation-reduction (i.e., redox) reactions provide the bridge to connect the molecular modalities of biology with the electronic modalities of devices. The Payne lab is developing new experimental methods (e.g., spectroelectrochemistry) to study redox-based communication. These methods are providing new insights on biological communication and suggest how redox-active biological materials such as melanin may interact with their environment (Figure 2).
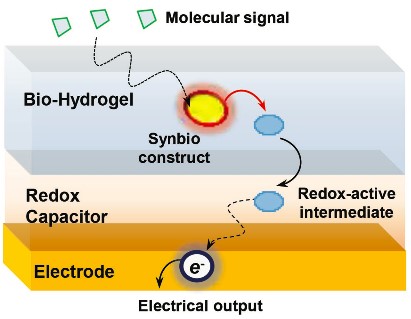
The lab is also developing signal processing methods that enable devices and biology to “talk” to each other through redox mechanisms (Figure 3). Coupling these methods with advances in synthetic biology (synbio) promises to offer transformative capabilities by enabling the integration of the information processing capabilities of biology into electronics. Specifically, synbio constructs are being engineered to transduce molecular information into electrical outputs for advanced sensing, while communication in the opposite direction is enabling electrical inputs to be transduced by other synbio constructs into molecular outputs that can alter biological behaviors.

Publications
- Scrambling Signal Modularity in Bottom-up Assembled Synthetic Pseudomonas Consortia Reveals Robust Information Transfer.
- Electrochemistry as a Tool for Redox-Based Bio-Information Processing.
- How Electric Field Remodels the Nanofibril Structure of Chitosan Hydrogels: the Role of Dewetting During Electro-Assembly.
- de novo Fabrication of Dense Collagen Matrices with Patterned Hierarchical Structures for Corneal Stromal Tissue Repair.
- Measuring oxidative stress by the iridium reducing capacity assay (Ir-RCA).
- 3D Printed Spectroelectrochemical Platform for Redox-Based Bioelectronics.
- Proline-Selective Electrochemiluminescence Detecting a Single Amino Acid Variation Between A1 and A2 β-Casein Containing Milks.
- Detecting features of antibody structure through their mediator-accessible redox activities.
- Biomimetic Redox Capacitor To Control the Flow of Electrons.
- Electrobiofabrication of antibody sensor interfaces within a 3D printed device yield rapid and robust electrochemical measurements of titer and glycan structure.
- Redox-mediated Biomolecular information transfer in single electrogenetic biological cells.
- Pilot study indicates that a gluten-free diet lowers oxidative stress for gluten-sensitive persons with schizophrenia.
- Redox active plant phenolic, acetosyringone, for electrogenetic signaling.
- Effect of Acetylation on the Nanofibril Formation of Chitosan from All-Atom De Novo Self-Assembly Simulations.
- Electro-Sorting Create Heterogeneity: Constructing A Multifunctional Janus Film with Integrated Compositional and Microstructural Gradients for Guided Bone Regeneration.
- Excite the unexcitable: engineering cells and redox signaling for targeted bioelectronic control.
- Redox-enabled electronic interrogation and feedback control of hierarchical and networked biological systems.
- Revealing Redox Behavior of Molybdenum Disulfide and Its Application as Rechargeable Antioxidant Reservoir.
- Electro-Biofabrication. Coupling Electrochemical and Biomolecular Methods to Create Functional Bio-Based Hydrogels.
- Electrogenetic signaling and information propagation for controlling microbial consortia via programmed lysis.
- Enhanced electrochemical measurement of β-galactosidase activity in whole cells by coexpression of lactose permease, LacY.
- Single Step Assembly of Janus Porous Biomaterial by Sub-Ambient Temperature Electrodeposition.
- Protein G: β-galactosidase fusion protein for multi-modal bioanalytical applications.
- Quorum sensing componentry opens new lines of communication.
- Network-based redox communication between abiotic interactive materials.
- Electrogenetic Signal Transmission and Propagation in Coculture to Guide Production of a Small Molecule, Tyrosine.
- Electro-assembly of a dynamically adaptive molten fibril state for collagen.
- Parsed synthesis of pyocyanin via co-culture enables context-dependent intercellular redox communication.
- Association of acute psychosocial stress with oxidative stress: Evidence from serum analysis.
- Mediated electrochemistry for redox-based biological targeting: entangling sensing and actuation for maximizing information transfer.
- A Redox-Based Autoinduction Strategy to Facilitate Expression of 5xCys-Tagged Proteins for Electrobiofabrication.
- Electronic signals are electrogenetically relayed to control cell growth and co-culture composition.
- Mediated Electrochemical Probing: A Systems-Level Tool for Redox Biology.
- Simple, rapidly electroassembled thiolated PEG-based sensor interfaces enable rapid interrogation of antibody titer and glycosylation.
- Interactive Materials for Bidirectional Redox-Based Communication.
- Bioelectronic control of a microbial community using surface-assembled electrogenetic cells to route signals.
- Redox Electrochemistry to Interrogate and Control Biomolecular Communication.
- Transglutaminase-mediated assembly of multi-enzyme pathway onto TMV brush surfaces for synthesis of bacterial autoinducer-2.
- Polyelectrolyte in Electric Field: Disparate Conformational Behavior along an Aminopolysaccharide Chain.
- A redox-based electrogenetic CRISPR system to connect with and control biological information networks.
- A Coculture Based Tyrosine-Tyrosinase Electrochemical Gene Circuit for Connecting Cellular Communication with Electronic Networks.
- Quorum Sensing Communication: Molecularly Connecting Cells, Their Neighbors, and Even Devices.
- Redox Is a Global Biodevice Information Processing Modality.
- Catechol-Based Capacitor for Redox-Linked Bioelectronics.
- Redox-Channeling Polydopamine-Ferrocene (PDA-Fc) Coating To Confer Context-Dependent and Photothermal Antimicrobial Activities.
- Melanin Produced by the Fast-Growing Marine Bacterium Vibrio natriegens through Heterologous Biosynthesis: Characterization and Application.
- Redox Activities of Melanins Investigated by Electrochemical Reverse Engineering: Implications for their Roles in Oxidative Stress.
- Tethered molecular redox capacitors for nanoconfinement-assisted electrochemical signal amplification.
- Pro- and Anti-oxidant Properties of Redox-Active Catechol-Chitosan Films.
- Validation of oxidative stress assay for schizophrenia.
- Erratum: Catechol-Based Hydrogel for Chemical Information Processing. Biomimetics 2017, 2, 11.
- Electrobiofabrication: electrically based fabrication with biologically derived materials.
- Coupling Self-Assembly Mechanisms to Fabricate Molecularly and Electrically Responsive Films.
- Reversibly Reconfigurable Cross-Linking Induces Fusion of Separate Chitosan Hydrogel Films.
- A platform of genetically engineered bacteria as vehicles for localized delivery of therapeutics: Toward applications for Crohn's disease.
- Selective assembly and functionalization of miniaturized redox capacitor inside microdevices for microbial toxin and mammalian cell cytotoxicity analyses.
- An immune magnetic nano-assembly for specifically amplifying intercellular quorum sensing signals.
- Radical Scavenging Activities of Biomimetic Catechol-Chitosan Films.
- Catechol-chitosan redox capacitor for added amplification in electrochemical immunoanalysis.
- Focusing quorum sensing signalling by nano-magnetic assembly.
- Electrodeposition of a magnetic and redox-active chitosan film for capturing and sensing metabolic active bacteria.
- Engineering bacterial motility towards hydrogen-peroxide.
- Signal processing approach to probe chemical space for discriminating redox signatures.
- Modification and Assembly of a Versatile Lactonase for Bacterial Quorum Quenching.
- The Role of Microsystems Integration Towards Point-of-Care Clozapine Treatment Monitoring in Schizophrenia.
- Electrical Programming of Soft Matter: Using Temporally Varying Electrical Inputs To Spatially Control Self Assembly.
- Connecting Biology to Electronics: Molecular Communication via Redox Modality.
- Spectroelectrochemical Reverse Engineering DemonstratesThat Melanin's Redox and Radical Scavenging Activities Are Linked.
- The Analgesic Acetaminophen and the Antipsychotic Clozapine Can Each Redox-Cycle with Melanin.
- Incorporating LsrK AI-2 quorum quenching capability in a functionalized biopolymer capsule.
- The Binding Effect of Proteins on Medications and Its Impact on Electrochemical Sensing: Antipsychotic Clozapine as a Case Study.
- Controlling localization of Escherichia coli populations using a two-part synthetic motility circuit: An accelerator and brake.
- The interplay of electrode- and bio-materials in a redox-cycling-based clozapine sensor.
- Catechol-Based Hydrogel for Chemical Information Processing.
- Molecular processes in an electrochemical clozapine sensor.
- Reliable clinical serum analysis with reusable electrochemical sensor: Toward point-of-care measurement of the antipsychotic medication clozapine.
- Electronic control of gene expression and cell behaviour in Escherichia coli through redox signalling.
- Electrochemical reverse engineering: A systems-level tool to probe the redox-based molecular communication of biology.
- Redox Probing for Chemical Information of Oxidative Stress.
- Using a Redox Modality to Connect Synthetic Biology to Electronics: Hydrogel-Based Chemo-Electro Signal Transduction for Molecular Communication.
- A Facile Two-Step Enzymatic Approach for Conjugating Proteins to Polysaccharide Chitosan at an Electrode Interface.
- Modular construction of multi-subunit protein complexes using engineered tags and microbial transglutaminase.
- Fusing Sensor Paradigms to Acquire Chemical Information: An Integrative Role for Smart Biopolymeric Hydrogels.
- Constructing "quantized quorums" to guide emergent phenotypes through quorum quenching capsules.
- Data on biochemical fluxes generated from biofabricated enzyme complexes assembled through engineered tags and microbial transglutaminase.
- Conferring biological activity to native spider silk: A biofunctionalized protein-based microfiber.
- Electro-molecular Assembly: Electrical Writing of Information into an Erasable Polysaccharide Medium.
- Electrochemical Probing through a Redox Capacitor To Acquire Chemical Information on Biothiols.
- Probing Energy and Electron Transfer Mechanisms in Fluorescence Quenching of Biomass Carbon Quantum Dots.
- Paraquat-Melanin Redox-Cycling: Evidence from Electrochemical Reverse Engineering.
- Networking biofabricated systems through molecular communication.
- Electrochemical Fabrication of Functional Gelatin-Based Bioelectronic Interface.
- Reverse Engineering Applied to Red Human Hair Pheomelanin Reveals Redox-Buffering as a Pro-Oxidant Mechanism.
- Electrochemical Measurement of the β-Galactosidase Reporter from Live Cells: A Comparison to the Miller Assay.
- Functionalizing Soft Matter for Molecular Communication.
- Nano-guided cell networks as conveyors of molecular communication.
- pH-Responsive Self-Assembly of Polysaccharide through a Rugged Energy Landscape.
- A 'bioproduction breadboard': programming, assembling, and actuating cellular networks.
- Multidimensional mapping method using an arrayed sensing system for cross-reactivity screening.
- Bacterial secretions of nonpathogenic Escherichia coli elicit inflammatory pathways: a closer investigation of interkingdom signaling.
- Electrochemical study of the catechol-modified chitosan system for clozapine treatment monitoring.
- Rapid and repeatable redox cycling of an insoluble dietary antioxidant: electrochemical analysis.
- Electronic modulation of biochemical signal generation.
- Fabrication of biomimetically patterned surfaces and their application to probing plant-bacteria interactions.
- Information processing through a bio-based redox capacitor: signatures for redox-cycling.
- Context-dependent redox properties of natural phenolic materials.
- Redox-capacitor to connect electrochemistry to redox-biology.
- Materials science. Nature's other self-assemblers.
- Optically clear alginate hydrogels for spatially controlled cell entrapment and culture at microfluidic electrode surfaces.
- Autonomous bacterial localization and gene expression based on nearby cell receptor density.
- Reverse engineering to suggest biologically relevant redox activities of phenolic materials.
- Amplified and in situ detection of redox-active metabolite using a biobased redox capacitor.
- Biofabricated film with enzymatic and redox-capacitor functionalities to harvest and store electrons.
- Glucose oxidase-mediated gelation: a simple test to detect glucose in food products.
- Encapsulated fusion protein confers "sense and respond" activity to chitosan-alginate capsules to manipulate bacterial quorum sensing.
- Bridging the bio-electronic interface with biofabrication.
- Biofabrication of stratified biofilm mimics for observation and control of bacterial signaling.
- Electrodeposition of a biopolymeric hydrogel: potential for one-step protein electroaddressing.
- pH- and voltage-responsive chitosan hydrogel through covalent cross-linking with catechol.
- Integrated biofabrication for electro-addressed in-film bioprocessing.
- Light-activated ionic gelation of common biopolymers.
- Biocompatible multi-address 3D cell assembly in microfluidic devices using spatially programmable gel formation.
- Electroaddressing agarose using Fmoc-phenylalanine as a temporary scaffold.
- Biofabrication with biopolymers and enzymes: potential for constructing scaffolds from soft matter.
- Redox-cycling and H2O2 generation by fabricated catecholic films in the absence of enzymes.
- In situ generation of pH gradients in microfluidic devices for biofabrication of freestanding, semi-permeable chitosan membranes.
- Biomimetic sealant based on gelatin and microbial transglutaminase: an initial in vivo investigation.
- Chitosan-coated wires: conferring electrical properties to chitosan fibers.
- Biofabrication of antibodies and antigens via IgG-binding domain engineered with activatable pentatyrosine pro-tag.
- Towards area-based in vitro metabolic engineering: assembly of Pfs enzyme onto patterned microfabricated chips.
- Orthogonal enzymatic reactions for the assembly of proteins at electrode addresses.
- Design optimization for bioMEMS studies of enzyme-controlled metabolic pathways.
- Chitosan-coated electrodes for bimodal sensing: selective post-electrode film reaction for spectroelectrochemical analysis.
- Chitosan fibers: versatile platform for nickel-mediated protein assembly.
- Programmable assembly of a metabolic pathway enzyme in a pre-packaged reusable bioMEMS device.
- Chitosan biotinylation and electrodeposition for selective protein assembly.
- Protein assembly onto patterned microfabricated devices through enzymatic activation of fusion pro-tag.
- Transglutaminase crosslinked gelatin as a tissue engineering scaffold.
- Chitosan: a soft interconnect for hierarchical assembly of nano-scale components.
- Biopolymer-based materials: the nanoscale components and their hierarchical assembly.
- Magnetic nanofactories: localized synthesis and delivery of quorum-sensing signaling molecule autoinducer-2 to bacterial cell surfaces.
- Reversible vesicle restraint in response to spatiotemporally controlled electrical signals: a bridge between electrical and chemical signaling modes.
- Mechano-transduction of DNA hybridization and dopamine oxidation through electrodeposited chitosan network.
- Chitosan-mediated in situ biomolecule assembly in completely packaged microfluidic devices.
- Bioinspired vesicle restraint and mobilization using a biopolymer scaffold.
- Tyrosine-based "activatable pro-tag": enzyme-catalyzed protein capture and release.
- Transition from unilamellar to bilamellar vesicles induced by an amphiphilic biopolymer.
- Biofabrication with chitosan.
- Gelatin-based biomimetic tissue adhesive. Potential for retinal reattachment.
- Patterned assembly of genetically modified viral nanotemplates via nucleic acid hybridization.
- Chitosan-mediated and spatially selective electrodeposition of nanoscale particles.
- Thermo-biolithography: a technique for patterning nucleic acids and proteins.
- Signal-directed sequential assembly of biomolecules on patterned surfaces.
- Vesicle--biopolymer gels: networks of surfactant vesicles connected by associating biopolymers.
- Biofabrication: using biological materials and biocatalysts to construct nanostructured assemblies.
- Mechanical properties of biomimetic tissue adhesive based on the microbial transglutaminase-catalyzed crosslinking of gelatin.
- Enzymatic grafting of peptides from casein hydrolysate to chitosan. Potential for value-added byproducts from food-processing wastes.
- A robust technique for assembly of nucleic acid hybridization chips based on electrochemically templated chitosan.
- Ammonium effects on streptonigrin biosynthesis by Streptomyces flocculus.
- Improved production of heterologous protein from Streptomyces lividans.
- Development of a strategy to control the dissolved concentrations of oxygen and carbon dioxide at constant shear in a plant cell bioreactor.
- Genetic engineering approach to toxic waste management: case study for organophosphate waste treatment.
