Protein Structures Identified by IBBR Team Provide Insights into T Cell Recognition of Targets for Cancer Therapies
Tue, Aug 4, 2020
 August 4, 2020 – Adoptive T cell therapy (ACT) is a treatment for patients with several forms of cancer. This therapeutic effect is mediated by T cells through recognition of tumor neoantigens, which are proteins not previously recognized by the immune system. To better understand interactions between neoantigens and T cell receptors (TCRs) that could impact therapeutic development, a team in the laboratory of IBBR Investigator Dr. Roy Mariuzza, Professor, UMCP Department of Cell Biology and Molecular Genetics, in collaboration with IBBR Fellow Brian Pierce, Assistant Professor, UMCP Department of Cell Biology and Molecular Genetics and IBBR Fellow D. Travis Gallagher of the National Institute of Standards and Technology (NIST), recently published a study in the journal Nature Communications describing structural characterization of TCRs bound to tumor-specific neoantigens.
August 4, 2020 – Adoptive T cell therapy (ACT) is a treatment for patients with several forms of cancer. This therapeutic effect is mediated by T cells through recognition of tumor neoantigens, which are proteins not previously recognized by the immune system. To better understand interactions between neoantigens and T cell receptors (TCRs) that could impact therapeutic development, a team in the laboratory of IBBR Investigator Dr. Roy Mariuzza, Professor, UMCP Department of Cell Biology and Molecular Genetics, in collaboration with IBBR Fellow Brian Pierce, Assistant Professor, UMCP Department of Cell Biology and Molecular Genetics and IBBR Fellow D. Travis Gallagher of the National Institute of Standards and Technology (NIST), recently published a study in the journal Nature Communications describing structural characterization of TCRs bound to tumor-specific neoantigens.
Tumor neoantigens arise upon alterations of DNA in cells and the resulting proteins can drive cancer initiation and progression. ACT uses the body’s own immune cells to treat cancer. In ACT with genetically modified peripheral blood T cells, peripheral blood T cells are isolated from a patient. A novel TCR which recognizes tumor neoantigens is then introduced into these T cells. The modified T cells are then transferred back to the same patient to target the cancer cells.
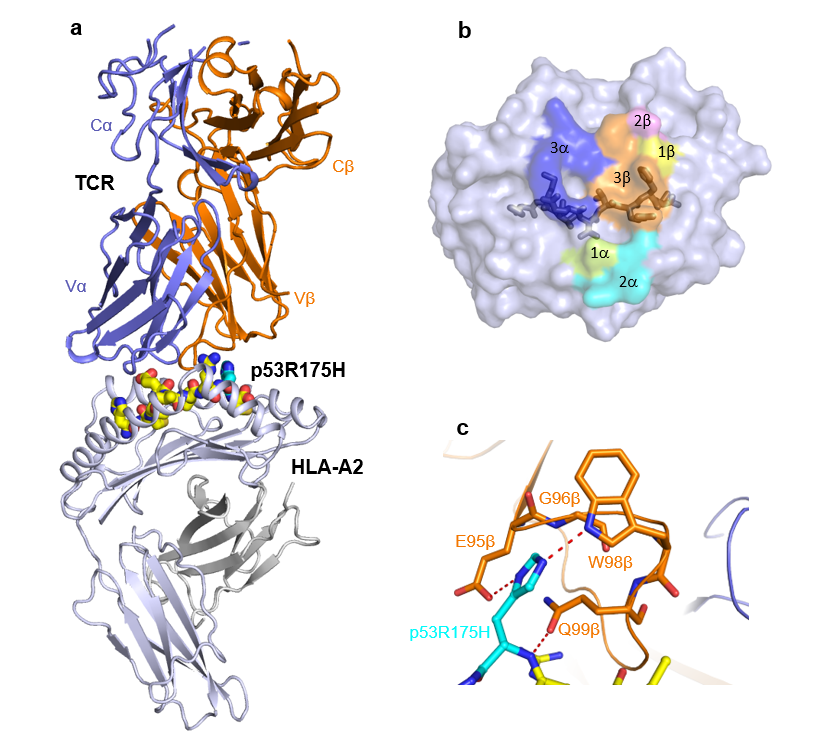
The research team studied a neoantigen from the p53 protein, which is a tumor suppressor that is the most frequently mutated gene across all cancer types. Mutations in p53 are associated with progression of malignancies and are therefore candidates of interest as targets for ACT. To better understand TCR recognition of cancer neoantigens, the group determined the crystal structures of three tumor-specific TCRs in complex with a p53 neoantigen (p53R175H) and a human MHC molecule (HLA-A2). One of these complexes is shown in the Figure. Since TCRs for ACT need to specifically recognize the neoantigen and not the non-mutated protein, this study sheds light on how different the neoantigen and the natural non-mutated protein need to be in order to evoke an immune response. This study examined such differences in p53 proteins and found that cancer neoantigens need to differ only slightly to elicit an immune response. These findings can aid in structure-guided efforts to engineer therapeutic TCRs with improved potency toward enhancing treatments for patients.
