Biomolecular Science and Engineering Research Areas
Most biologics on the market and under development are glycoproteins, and currently account for the majority of the top drugs by sales. Major products include monoclonal antibodies, enzyme replacement therapies, subunit vaccines, and many other products. The structure of the glycans on most of these drugs impact their safety and function. However, the glycans remain one of the most challenging parts of many biologics to study, optimize, and control.
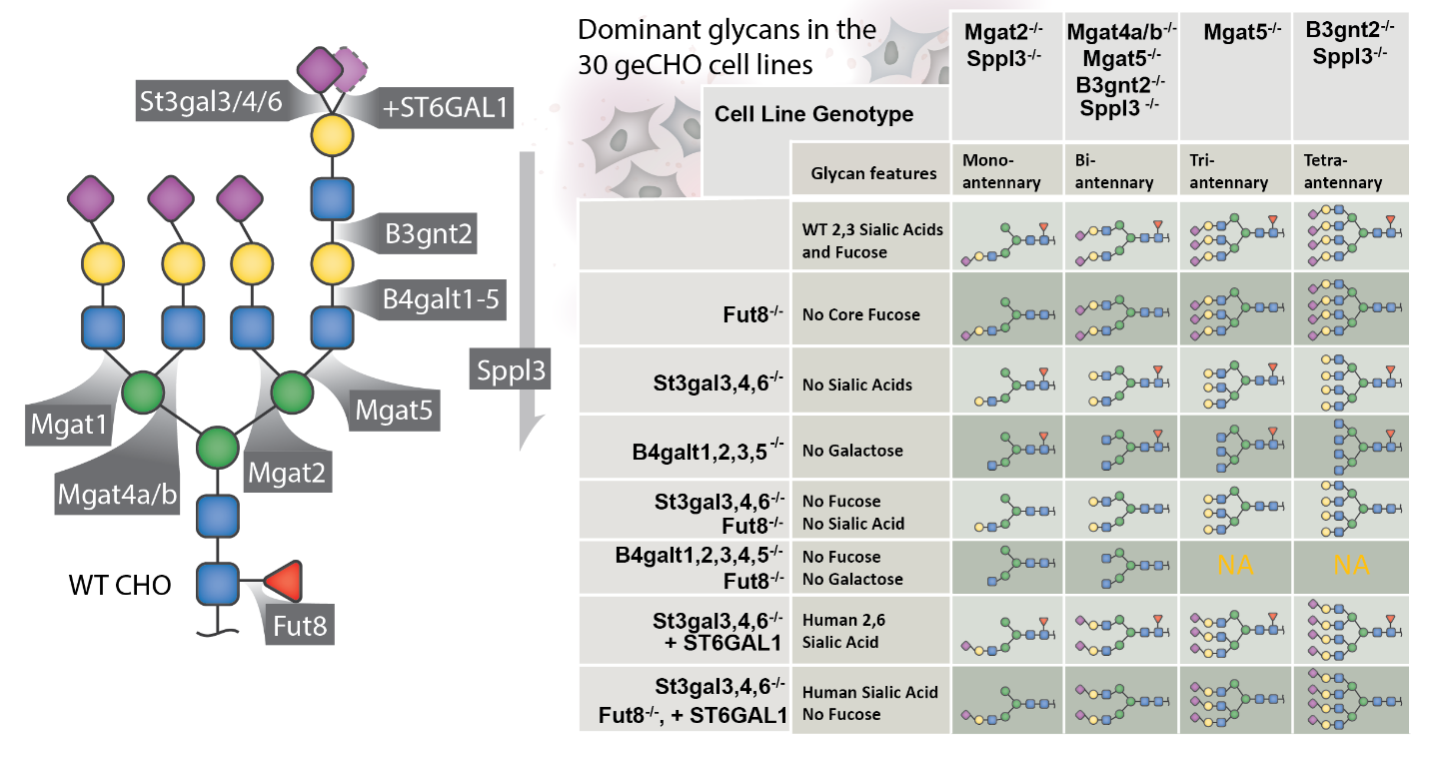
CGAB will leverage more than a decade of work in the development of CHO genomics, machine-learning, systems biology, and genome editing, all applied to optimize CHO cell factories and glycotherapeutics. We will expand the diversity of the geCHO cell panel (Fig. 9) and integrate synthetic biology circuits to enhance expression and control of the desired glycosylation on any product. To aid in process control, our systems biology and machine-learning models will be further refined for more precise in-line control during manufacturing runs. Thus, the work of the CGAB will harness and refine this difficult post-translational modification on leading biologics, making it a valuable design parameter in biomanufacturing.
Since the first monoclonal antibody therapeutics were approved two decades ago, there has been a transition to design molecules for optimal target recognition, pharmacokinetics, and therapeutic function. CGAB has an integrated team in protein engineering, structural biology, enzymology, and computational modeling focused on developing novel platform technologies.
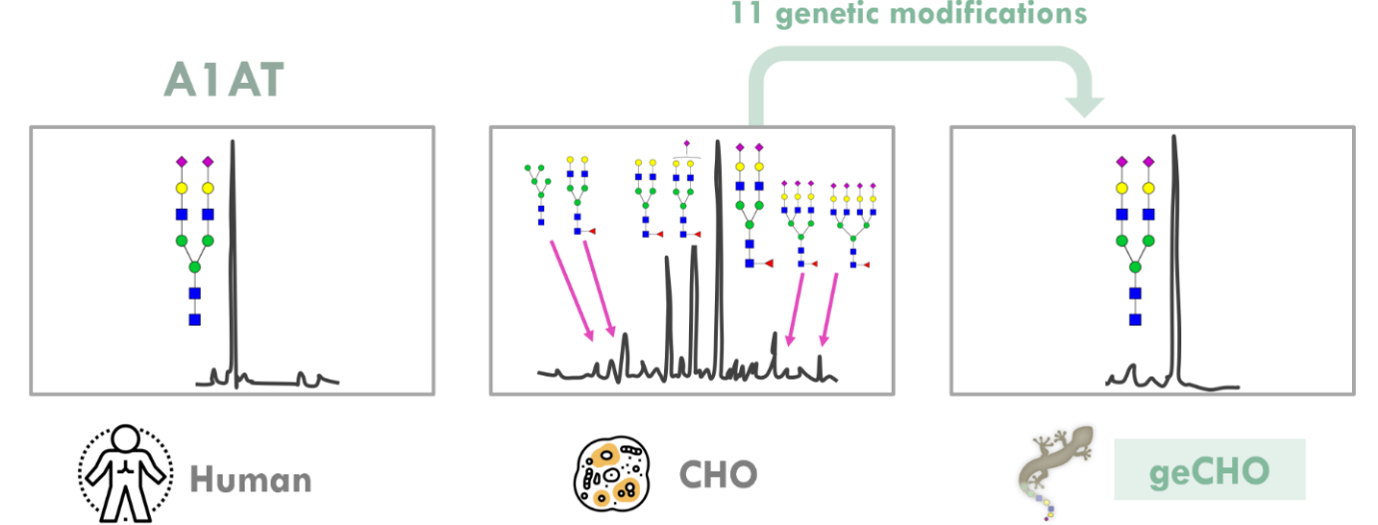
Several genetic disorders of the lung and blood are associated with mutations to serine protease inhibitors (Serpins), including alpha-1 antitrypsin (A1AT), antithrombin (AT), and C1-esterase inhibitor (C1-INH). Each of these SERPINs, when genetically deficient, is associated with hereditary syndrome with clinical manifestations, such as chronic obstructive pulmonary disease (A1AT), venous thromboembolism susceptibility (AT), and hereditary angioedema (C1-INH). These conditions can be treated using SERPIN augmentation therapy. Current therapies rely upon augmentation of the plasma-derived proteins, but plasma fractionation carries a risk of blood borne pathogens and prions, and is fraught with plasma supply chain bottlenecks. Efforts to produce these proteins recombinantly have resulted in products with reduced half-life and/or changes to protein function, and it seems that the glycan structures on these proteins underlie the changes. Using our geCHO technology, we can deploy a platform that can recombinantly produce therapeutic glycoproteins with native glycosylation (Fig. 10) for use in SERPIN augmentation therapy.
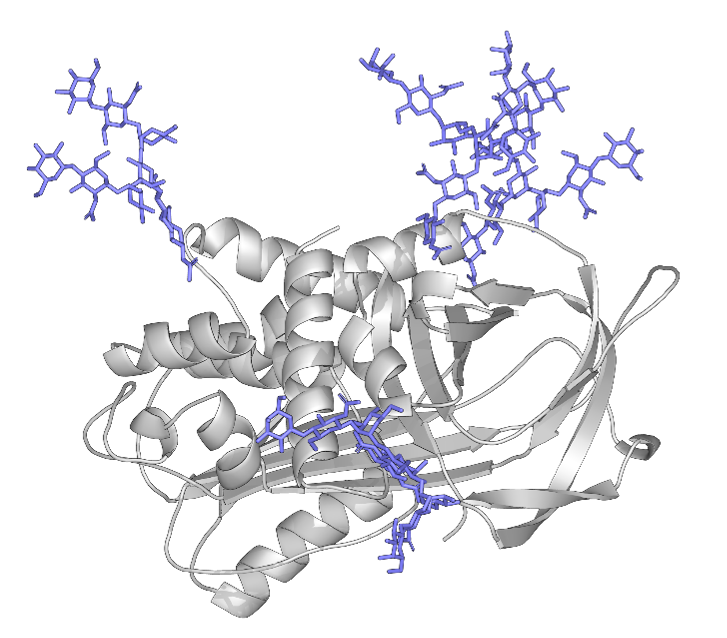
Another avenue of research that can leverage the geCHO technology is protein design optimization of advanced biomolecule therapeutics (Fig. 11). Toward that end, we have a comprehensive protein engineering pipeline that encompasses structure-based design optimizations, maximizing production yield and purity, and optimizing glycan content for maximum efficacy. This pipeline is applicable to a wide range of therapeutics currently under study/development at the Consortium. For example, plasma-derived proteins lack the opportunity for protein engineering-based improvements to protein potency or half-life. Thus, the ability of the geCHO platform to produce these proteins recombinantly affords us the opportunity to make structure-based optimizations to enhance potency by eliminating vulnerabilities in the target proteins, e.g. replacing oxidation-sensitive methionines, removing protease cleavage sites, or stabilizing key interfaces and domains. Moreover, since the geCHO platform contains a panel of 30 GMP-ready geCHO cells that will produce recombinant proteins with a diverse range of homogeneous glycans, we can use this technology to determine the optimal glycoform of each plasma-derived protein. In this regard, we are using glycan engineering as an additional method for protein engineering. In addition to SERPINs, other targets under development in this pipeline are butyrylcholinesterase, an important biodefense agent, and a number of vaccine antigens.
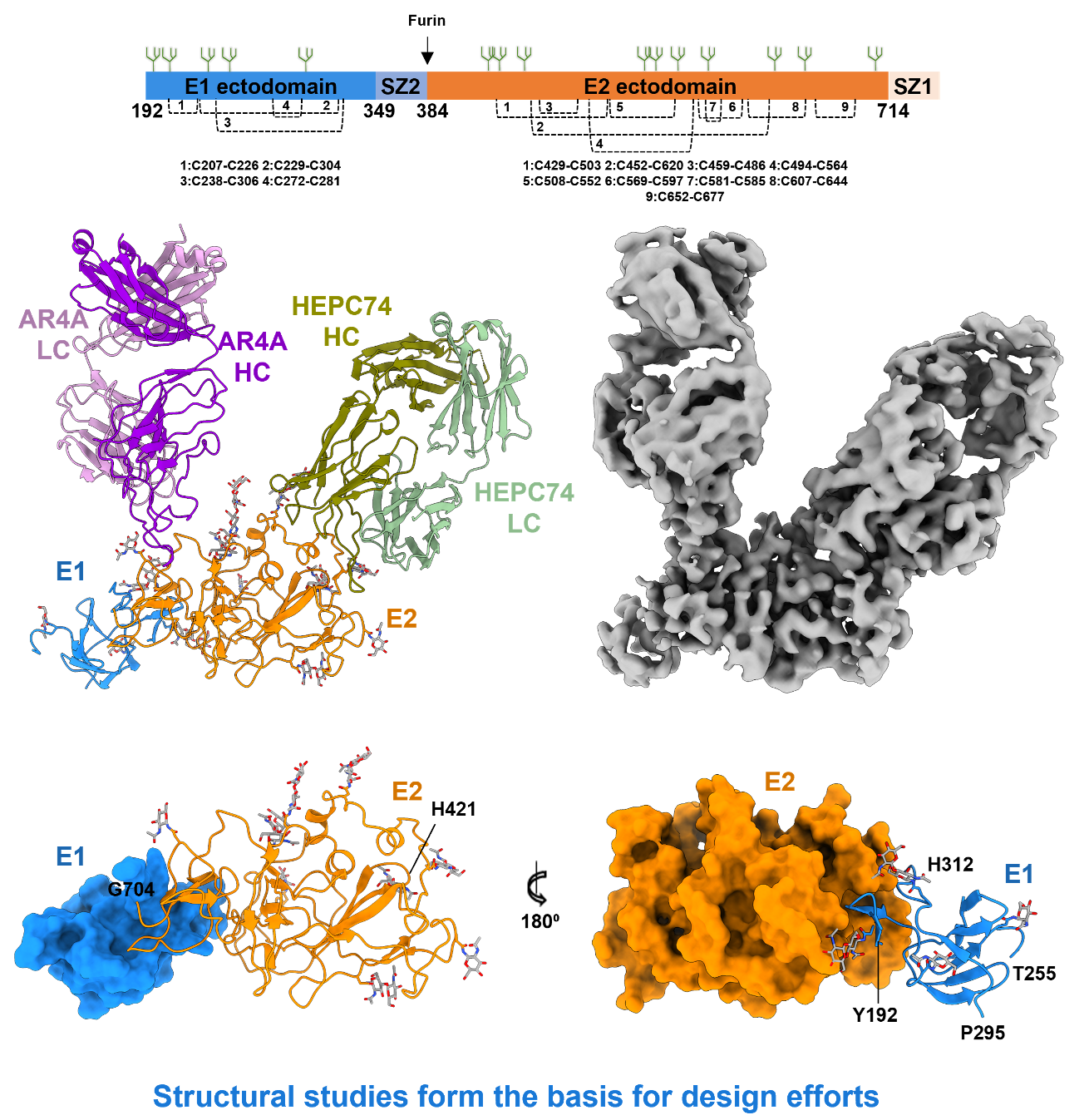
A better understanding of disease pathogenesis and the mechanisms that host cells mount in defense is key to the diagnosis, treatment, and prevention of disease. The CGAB leverages several decades of experience studying protein biomolecular structure and function, combining biochemistry, computational biology, and structural studies (NMR, X-ray crystallography, and more recently cryo-EM) to address these real-world issues. For example, many so called “vaccine-difficult” pathogens have co-evolved strategies to stimulate immune responses to epitopes not associated with protective immunity. The CGAB has an integrated team in virology, immunology, protein chemistry, molecular biology, structural biology, microbiology, computational analysis, and clinical medicine for developing predictive and experimental tools to advance the concept of “rational antigen design” to focus the immune system on conserved, conformational epitopes associated with protective immunity using hepatitis C virus (Fig. 12), HIV, Ebola virus (Fig. 13), and Zika virus as model systems.
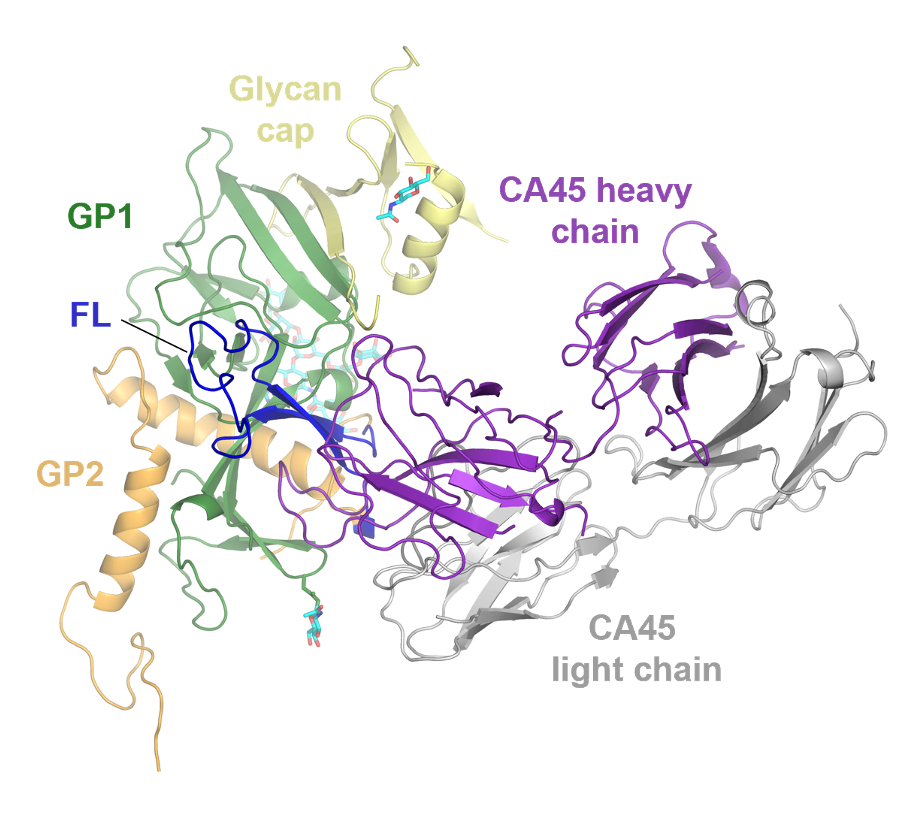
CGAB’s structure-based vaccine design (SBVD) program, complemented with a novel immunoadjuvant and delivery (IAD) system (see below), is focused on controlling antigenicity at the atomic-level to create immunogens capable of eliciting robust neutralizing and protective immune responses. A fundamental aspect of this paradigm involves the study of immune responses in infected individuals and vaccinated subjects to define antigenic targets of both protective and non-protective antibodies. The ability to identify and isolate antibodies from infected individuals has advanced in recent years through use of single B-cell cloning technologies combined with next generation sequencing analyses that define the kinetics and maturation pathways of related antibody gene clusters.
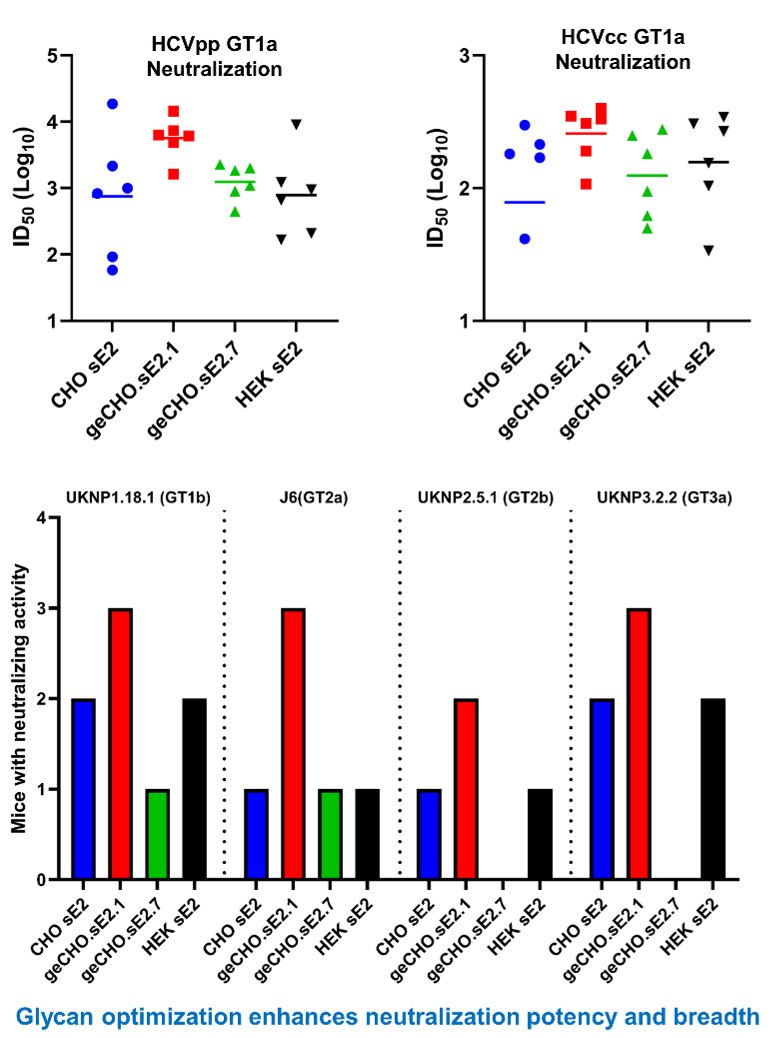
Glycoengineering has been relatively unexplored in vaccine design, despite evidence that glycans on viral antigens impact immune responses. Our geCHO system is a unique resource allowing precise control of glycosylation. It provides nearly homogenous glycans, allowing us to conduct a systematic assessment of glycan structure on vaccine potency and thus find which glycans most influence antigen properties and the development of the immune response. Standard eukaryotic expression systems preclude tight control of glycans on produced antigens, making analyses of how glycans impact immune recognition intractable. Moreover, recombinant glycoproteins typically include a heterogeneous mix of optimal glycans with suboptimal, deleterious, and/or absent glycans, introducing variability into the immune response and hampering efforts to study the roles of specific glycans in immune responses. Studies with influenza hemagglutinin, SARS CoV-2 spike, and HIV env have shown that glycan trimming to a single GlcNAc can favorably impact immunogenicity. The geCHO panel provides a resource to fine tune the glycan content of a vaccine antigen to optimize both the antibody (Fig. 14) and cellular immune responses. The glycan optimization approach is more powerful and systematic than enzymatic trimming and is used in concert with our SBVD efforts at CGAB. This combined approach provides clients and collaborators with technologies that are unique to the Consortium.
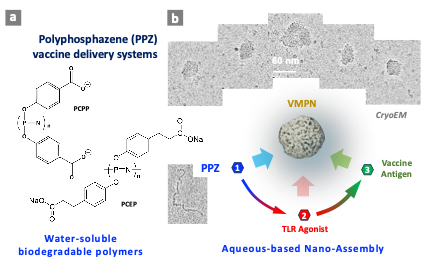
Polyphosphazene (PPZ) vaccine delivery and immunoadjuvant technology, which is currently being advanced at IBBR-UMD, is proven to dramatically improve immune responses to diverse viral and bacterial vaccine antigens. PPZ adjuvants are water-soluble synthetic macromolecules with phosphorus-nitrogen backbone and organic side groups, which can degrade in the body resulting in benign products. The key advantages of PPZ adjuvants are accelerated onset of the immune response, prolonged immunity, vaccine antigen dose sparing capabilities, and ability to modulate quality of the immune response. The lead PPZ product candidate – PCPP has shown excellent safety profile in five clinical trials, was produced under current Good Manufacturing Practice (GMP) conditions in millions human doses, and its Drug Master File was successfully registered with FDA. Recently, several important developments promoted this technology to a new level. CGAB researchers established that PPZ adjuvants spontaneously self-assemble with vaccine antigens resulting in nano-scale supramolecular structures that resemble viruses - Virus-Mimicking Polyphosphazene Nanoassemblies (VMPNs; Fig. 15). VMPNs can present not only multiple copies of the same antigenic protein, but create multi-protein assemblies, which is critical when the role of each immunogenic protein is yet to be defined. The group also developed an approach, which allows non-covalent assembly of various Toll-Like Receptor (TLR) agonist Immunomodulators (TLR 4, TLR 7/8) on VMPNs into a multimeric form (PPZ-R). Those include immunomodulators, such as resiquimod (R848), which is critical in inducing efficient responses against RNA based viruses, but proved to show suboptimal performance in clinical environment due to fast renal clearance and its dissociation from the antigen. We already demonstrated that PPZ-Rs overcome these challenges in vivo when administered with the antigen. These novel immunoadjuvants currently under investigation and preclinical development for HCV, Ebola virus, Zika virus, and Nipha virus vaccine candidates.
Lysosomal storage diseases (LSDs) are a group of ~60 inherited disorders affecting 1 in 5,000 newborns. LSDs are mostly due to defects in hydrolytic enzymes that reside within lysosomes, leading to unwanted and toxic buildup of waste products in cells. Supplementing defective enzymes by enzyme replacement therapy (ERT) is the most accepted treatment and a "universal" approach that can be applied regardless of the LSD mutation, disease, and effect. ERT is clinically available for 8 out of 60 LSDs and in clinical trials for four other LSDs. However, ERT is limited by the inability to efficiently (a) target enzymes across the blood brain barrier (BBB) to the central nervous system (CNS), precluding its use for most neurologic LSD patients, (b) deliver required enzyme amounts to other “hard-to-reach” organs, such as the lungs and bones, and (c) production of the optimal enzyme glycoform that is key to its proper structural folding and function. Glycan content of enzymes used for lysosomal ERT can in general affect their pharmacokinetics and biodistribution at the cellular and organ levels as well as intra-lysosomal degradation by proteases. One of CGAB’s targets is acid sphingomyelinase (ASM), which is used in the treatment of Niemann-Pick disease (NPD). Current treatments for NPD focus on non-neurologic symptoms of NPD, but CGAB has a form of ASM that more effectively targets recombinant ASM to the brain (Fig. 16).
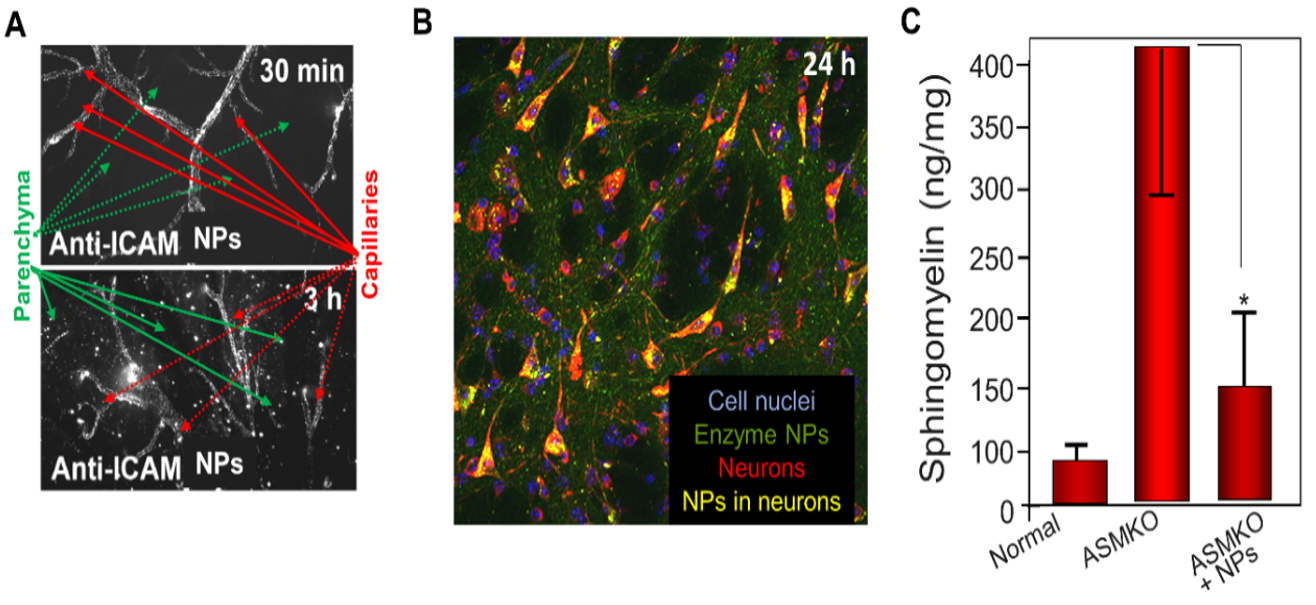
Glycans are important for proper ASM function, as removal of the some glycans leads to rapid cleavage and/or enzymatically dead proteins, along with aggregation in the endomembrane system. For another target, alpha-galactosidase A, production in different geCHO lines dramatically altered its biodistribution. Thus, we can leverage the geCHO platform to produce glycoforms that optimize the activity, half-life and biodistribution of targets for lysosomal ERT.
An emerging approach to therapeutics involves harnessing the power of human immune cells to fight disease. Key to this approach is a better understanding of T cell biology. Structural and computational biologists at the CGAB are exploring the structure and function of T cell receptors (TCRs), glycoproteins involved in both recognition and signaling by the immune system. CGAB researchers are using X-ray crystallography, NMR spectroscopy, and directed evolution to probe the structure and dynamics of the complex formed between TCRs and the important immune signaling receptor CD3. Also, a web server called TCRmodel has been developed which creates high-resolution structural models of TCRs in complex with peptide-Major Histocompatibility Complex (pMHC) targets from sequence. The goal of this work is to enhance understanding of T cell immunity, opening the door to new and better immunotherapies for cancers, as well as autoimmune and infectious diseases.
Glycosylation is relevant for TCR recognition and function. Both TCR and CD3 proteins contain N-linked glycans, as well as antigen-presenting major histocompatibility complex (MHC) and CD1 proteins. In some cases, peptide antigens targeted by TCRs are glycosylated. A better understanding of the molecular and structural basis of N-glycosylation in TCR recognition, and potential utilization of these insights to engineer and improve T cell therapeutics, is an important goal.
Most biologics suffer from suboptimal pharmacokinetics with poor access to their targets in the body, which also precludes them from being administered via non-invasive routes. This hinders safety and success of current treatments, and future expansion of this new pharmaceutical niche. CGAB is developing innovative delivery and stabilization technologies to enhance the efficacy of protein therapeutics (antibodies and enzymes), vaccines, and diagnostic products (Fig. 17).
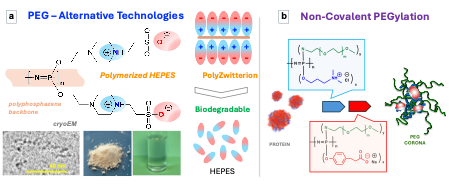
The creation of a highly hydrated dynamic steric barrier around protein and peptide therapeutics reduces their undesirable recognition by the host immune system. This, in turn, minimizes their intrinsic immunogenicity and a rapid clearance – well-known shortcomings of advanced macromolecular drugs and nanomedicines. Current strategies in the formation of hydrated steric (stealth) barriers on surfaces or around nanomedicines continue to rely on PEGylation - a commercially successful technology, which is based on covalent modification of a substrate with poly(ethylene glycol)(PEG). However, recent studies on the emergence of anti-PEG antibodies pose reasonable concerns over further advancement of this strategy and intensified the search for alternative technologies. Zwitterionic polymers – ampholytic macromolecules containing ionic moieties of opposite sign on the same pendant groups, exhibit strong protein-repulsive properties and an inherent biological inertness. For that reason, these highly hydrated inner salt macromolecules have emerged as some of the most viable alternatives to PEGylation. However, the structural diversity of polymer zwitterions remains limited and currently available macromolecules do not possess an intrinsic ability to undergo hydrolytical degradation – an important prerequisite for use in drug delivery applications. The CGAP group synthesized a zwitterionic polymer - a multimerized form of a widely used, biologically benign buffering agent - 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES), which is covalently assembled on a hydrolytically degradable polyphosphazene backbone. The resulting high molar mass polymer - polyHEPES-pz contains approximately two thousand pendent HEPES moieties per macromolecular chain and shows excellent solubility in aqueous solutions. Direct visualization of this polymer within the vitrified state using cryogenic electron microscopy (cryoEM) reveals individual chains of linear macromolecules. The polymer exhibits typical polyzwitterionic solution behavior, such as the “salting-in” effect and upper critical solution temperature (UCST) type miscibility profile. PolyHEPES-pz displays a pH and temperature dependent hydrolytic degradation pattern, demonstrates excellent in vitro compatibility with human red blood cells and strong resistance to interactions with plasma proteins – features, which highlight its utility for life sciences applications.
Another approach, which has been advanced by CGAB scientists, is based on the development of biodegradable PEGylated polyelectrolytes. These ionic macromolecules can self-assemble with proteins in aqueous formulations in a controlled fashion thereby creating a protective “hydration shield” via non-covalent interactions. This eliminates the need to perform laborious, expensive and potentially harmful chemical manipulations and extends the range of proteins suitable for further development. Stabilized proteins can be then further functionalized to modulate cell-surface interactions and enable cytosolic delivery and targeting of specific types of cells, such as cancer cells. CGAB researchers have been on the forefront of development of ‘non-covalent’ biodegradable PEGylation technology concept and engineering ‘smart’ macromolecular carriers for targeting various intracellular compartments.